 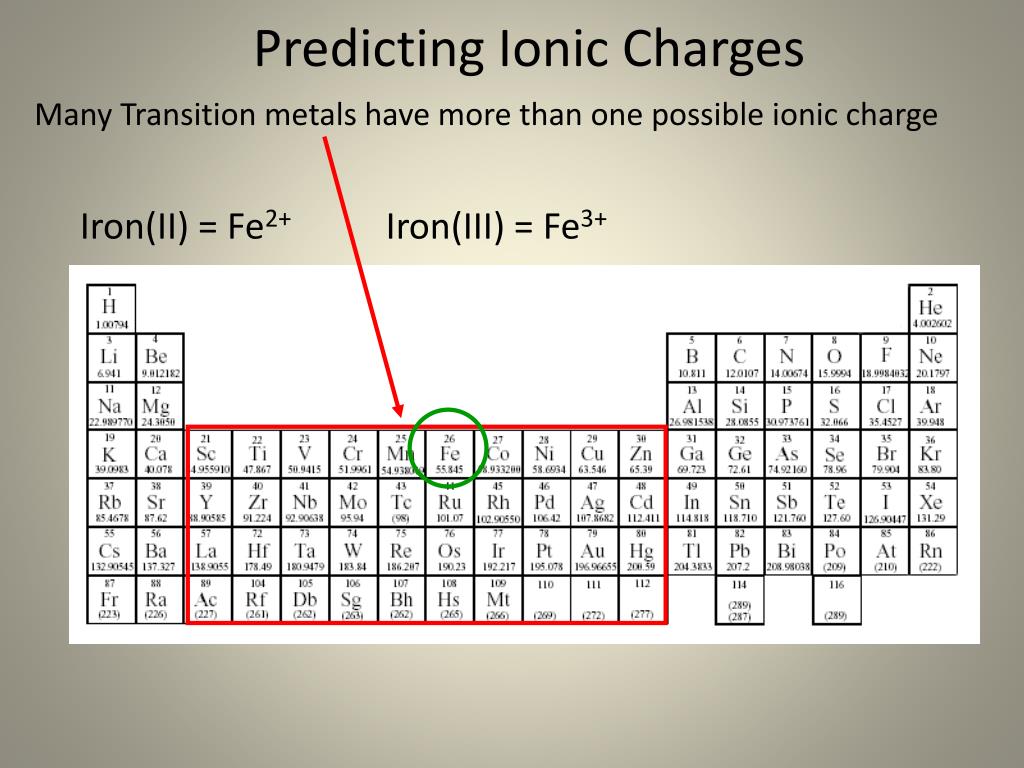
In addition, two of the ten most common compounds in the earth's crust are the two common oxides of iron, \(FeO\) and \(Fe2O3\). It is a very common element, fourth most abundant in the earth's crust. While these names are distinctive and, therefore, are considered chemically-correct, the common system has two significant limitations. Iron, which takes its English name from the old Anglo-Saxon and its symbol from the Latin, ferrum, was identified and used in prehistoric times. For example, the Latin stem for "iron" is "ferr-." Therefore, using the common system, Fe +2 is called the "ferrous ion," and Fe +3 is named as the "ferric ion." In most cases, the stem, or main portion, of the elemental name used in the common system is typically derived from the element's Latin name. FeCl 2 crystallizes from water as the greenish tetrahydrate, which is the form that is most commonly encountered in commerce and the. Iron-54 Metal Isotope 54Fe bulk & research qty manufacturer. The compound is white, but typical samples are often off-white. It is a paramagnetic solid with a high melting point. The "-ous" suffix indicates the cation with the lesser relative cation charge, and an "-ic" suffix is used to refer to a cation with a larger charge. Iron (II) chloride, also known as ferrous chloride, is the chemical compound of formula FeCl 2. The second system, called the common system uses two suffixes, "-ous" and "-ic," to distinguish the names of transition metal elements.\): Possible Transition Metal Charges and Their IUPAC Roman Numeral Designations Charge 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
